Shortened INCI lists and reduced preservatives push stability risk toward packaging interfaces. Airless pumps and narrow orifices buy time by limiting oxygen ingress and finger contact, but they do not replace formulation discipline or GMP.

Low-volume actives, water-light emulsions, and SKUs sold to consumers who dip fingers historically benefit from airless approaches. Match the pump elastomers and internal metallurgy to your pH and salt load.
If you are weighing a tube-style airless against a jar with a spatula, model the cumulative oxygen and microbial exposure over twenty real dispensing events—not a single laboratory squeeze. The difference often justifies tooling spend that looked excessive on spreadsheet day one.
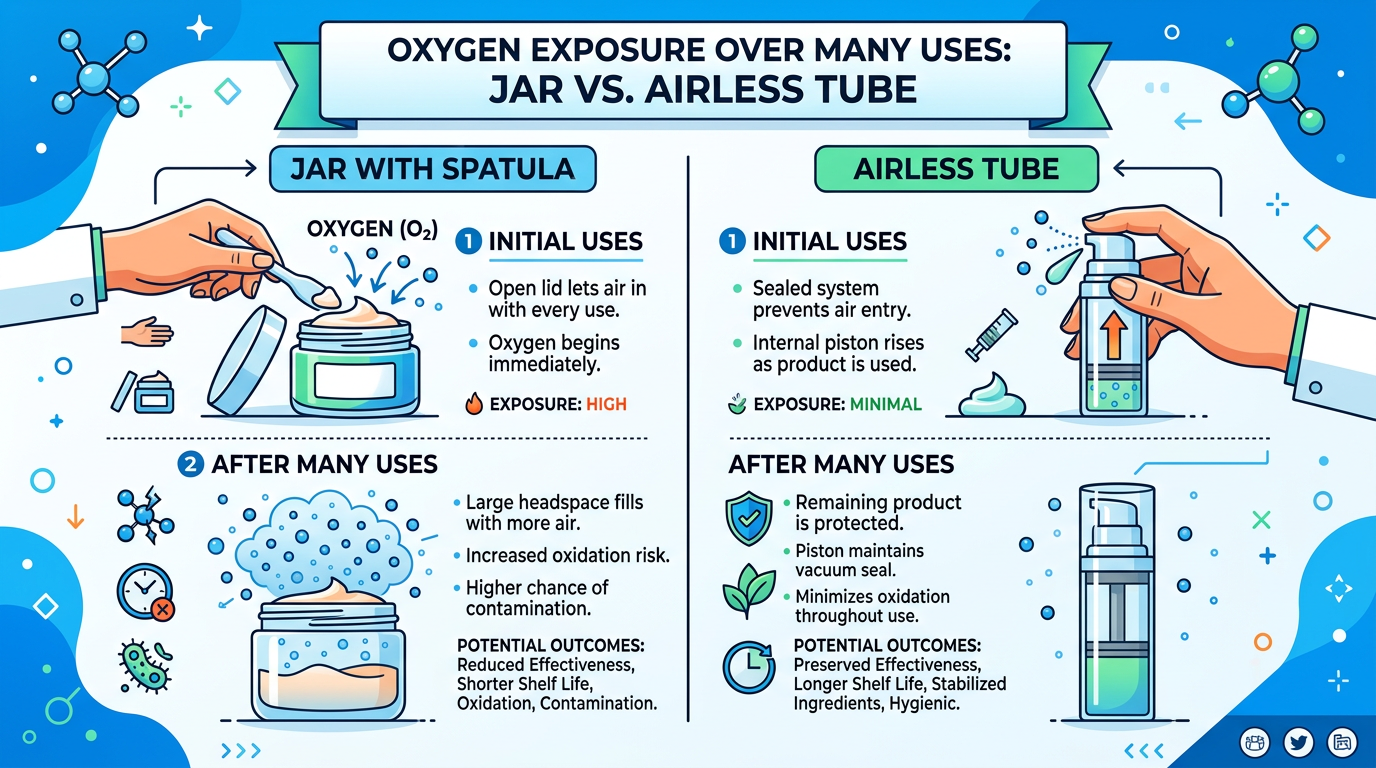
Challenge testing must include the chosen pack at production scale. Changing from a jar to an airless system is a formula change event, not a label tweak.
Document fill-line sanitization, downtime procedures, and cap torque drift across shifts. Packaging reduces risk; it does not sterilize a compromised batch.
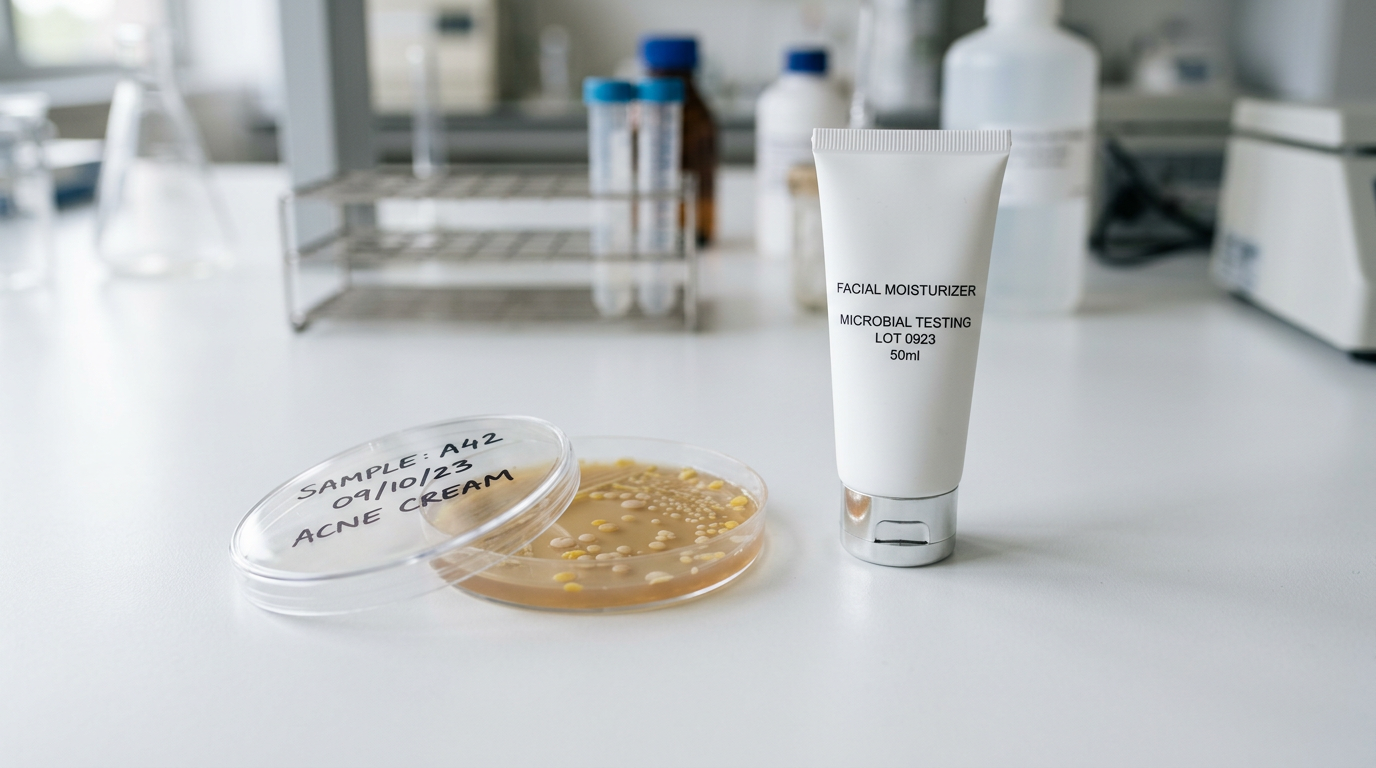
Use airless strategies where they measurably reduce risk. Skip them where they add cost without extending shelf life—consumers reward honesty more than hardware for its own sake.


 Add: No.2, Nanyu Street, Xinzao Town, Panyu District, Guangzhou City, China
Add: No.2, Nanyu Street, Xinzao Town, Panyu District, Guangzhou City, China
 Mobile:+86 13560168895
Mobile:+86 13560168895
 Email: [email protected]
Email: [email protected]
 WhatsApp: +86 13560168895
WhatsApp: +86 13560168895
 LEAVE A MESSAGE
LEAVE A MESSAGE




 Free Sample
Free Sample